Overview
Senior Investigator
Research Areas (IRP Lab Groups)
Mammalian skin is inhabited by various microbial communities known collectively as the microbiota. The host provides a home and nutrients for the microbes, and the microbes are crucial for stimulating healthy immunity, representing a mutually beneficial relationship. When this relationship changes, it may lead to perpetuating inflammation.
The Cutaneous Leukocyte Biology Section aims to understand how skin functions as an immune organ and studies mechanisms that underlie host-microbial symbiosis during health and disease, in both experimental models and human diseases.
Mechanisms of immune regulation and host-microbial symbiosis in the skin
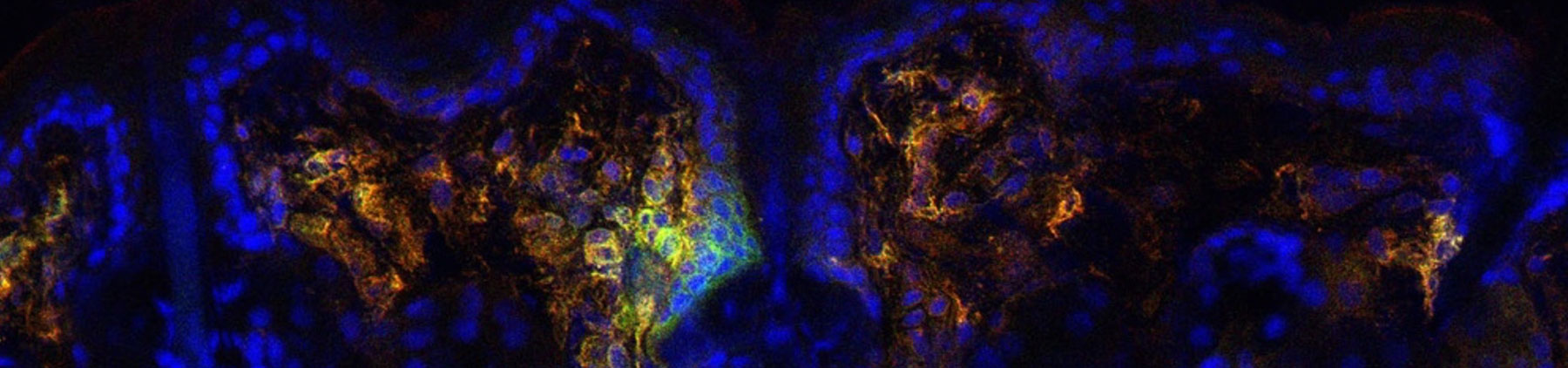
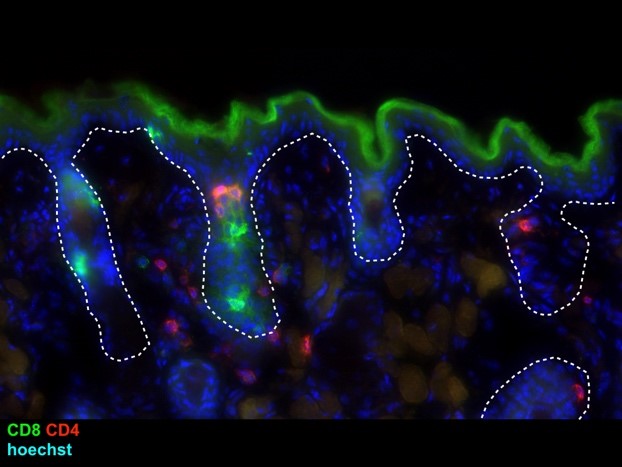
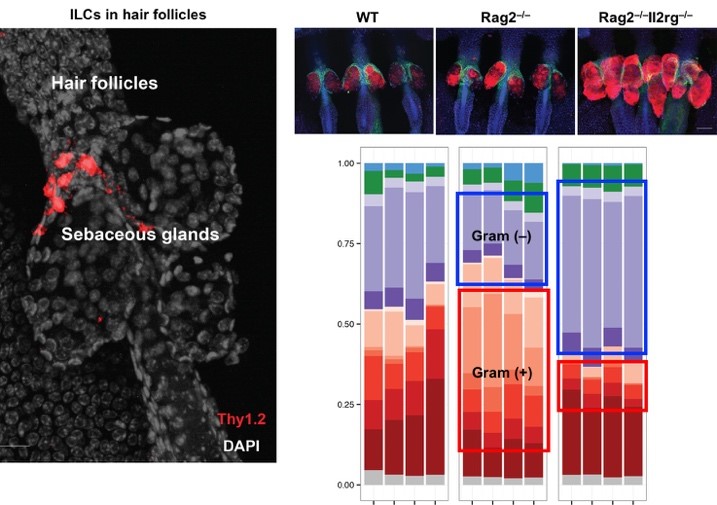
Hair follicles are fascinating structures that produce hair. The laboratory has established hair follicles as immunologically functional structures that recruit and control the localization of immune cells through the production of chemokines and cytokines. In turn, immune cells, such as the innate lymphoid cells, control sebaceous gland function to tune the balance of the microbiota during homeostasis.
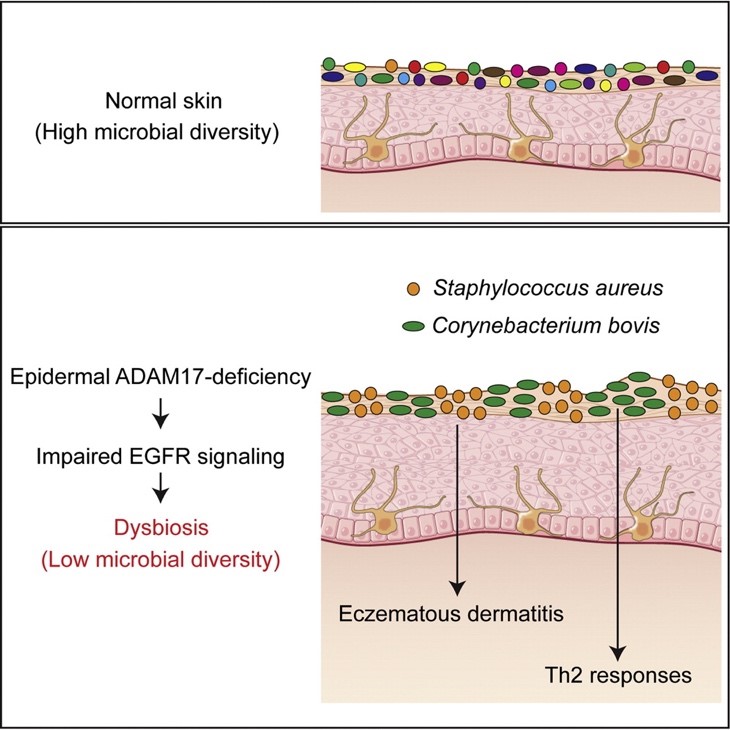
The laboratory utilizes a variety of experimental models to understand how host-microbe relationships are maintained and disrupted. In particular, the laboratory has generated a mouse model for atopic dermatitis that recapitulates the susceptibility of atopic dermatitis patients to skin colonization of Staphylococcus aureus. While the contribution of S. aureus in eczematous inflammation has long been debated, the laboratory has demonstrated S. aureus to be a crucial driver of inflammation.
Dissecting pathological inflammation directly in human diseases
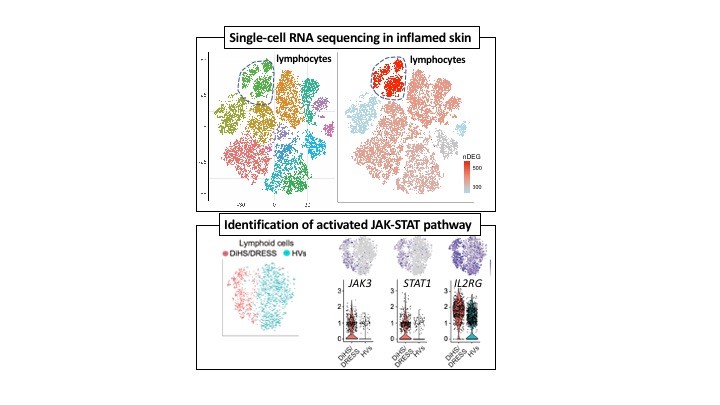
Advances in sequencing technologies, such as single-cell RNA sequencing, has enabled us to understand transcriptome at the single-cell level. These technologies provide us with opportunities to understand human diseases at unprecedented resolutions. The laboratory utilizes single-cell RNA sequencing and bioinformatics approaches to understand disease pathophysiology directly in humans and to understand how the immune system regulates the microbiota. Diseases of interest include, but are not limited to, severe drug hypersensitivities and primary immunodeficiencies.
Scientific Advances
Core Research Facilities
Labs at the NIAMS are supported by the following state-of-the-art facilities and services:
Staff

Image & Media Gallery
Career Opportunities
Stephen I. Katz Scholars in Dermatologic Research
We are seeking individuals interested in a career in dermatology clinical and translational research. Trainees who have completed accredited rheumatology or dermatology training programs are eligible to apply for this program. Applications will be considered on a rolling basis.
Postdoctoral Fellows
Postdoctoral fellows interested in integrating bioinformatics and immunology to study basic and translational skin immunology should send a cover letter, curriculum vitae with bibliography, and the names and contact information for three references to Dr. Nagao.